When Life Gets Loud, Your Body Is Worth Listening To: Know the Signs of Endometrial Cancer
(BPT) - One Woman's Story Highlights Why Postmenopausal Bleeding Should Never Be Ignored
Sponsored by GSK. Schari is a patient with endometrial cancer who will be discussing her experience; individual experiences may vary. Schari has been compensated by GSK for her participation in this article.
Cancer doesn't wait for the right moment, and it doesn't always announce itself loudly. Even when you're paying attention, symptoms can come and go or feel less urgent than everything else life throws your way.
For Schari, this is exactly what happened.
A few years after she entered menopause - typically defined as 12 straight months without a period - she experienced unexpected vaginal bleeding. Alarmed, she called her doctor and underwent testing. The results showed a thickened endometrial lining in her uterus, but she was told there was no immediate cause for alarm. The bleeding stopped, and life moved on.
She experienced abnormal bleeding twice more. The second time, her doctor performed a biopsy and found a nodule that turned out to be benign. Four years later, the bleeding returned again - but by then, Schari was dealing with the aftermath of a serious fall that reignited intense back pain. Compared to the pain she was experiencing, the bleeding felt like something that could wait.
"I remember thinking, 'The bleeding is probably nothing. This has happened before, and my tests were benign.' I was lulled into thinking this time it would be more of the same," she said.
Months later, she returned to her gynecologist for an exam, ultrasound, and biopsy. Schari remembers she was sitting in the bath when her doctor called with unexpected news: the findings showed endometrial carcinosarcoma, an aggressive type of endometrial cancer. She had recently gotten engaged to her partner, Patrick, and was busy planning their life together. The thought struck her in an instant: would she live to see her wedding, let alone the rest of her life plans?

Looking back, Schari sees that phone call as a turning point - and wonders how different things might have been if she had gotten an answer sooner. Her experience reflects a reality many women face: quieter symptoms can be overshadowed by bigger, louder problems. It's also a reminder of what postmenopausal bleeding can signal.
What Is Endometrial Cancer?
Endometrial cancer is a type of uterine cancer. It starts in the inner lining of the uterus, known as the endometrium. Uterine cancer and endometrial cancer are often used interchangeably, since endometrial cancer makes up the majority - about 90% - of cancers in the body of the uterus. Endometrial cancer is the most commonly diagnosed type of cancer that affects the gynecologic organs in the United States and it is estimated that more than 68,000 people will be diagnosed in 2026.
Common symptoms of endometrial cancer can include unusual vaginal bleeding or spotting, other vaginal discharge, pelvic pain, and unexplained weight loss. Because there is no routine screening test for endometrial cancer, regular gynecologic checkups are especially important - even after menopause. Any vaginal bleeding after menopause is never considered normal and should always be evaluated.
A family history of endometrial cancer increases one's risk, but women with no family history can also develop it. Additional risk factors for endometrial cancer can include obesity, polycystic ovary syndrome (PCOS), advancing age, and the use of postmenopausal estrogen therapy without progestin.
A Diagnosis - and a New Era of Treatment
After her diagnosis, Schari's gynecologic oncologist outlined an aggressive treatment plan. Surgery would involve removing her uterus, cervix, ovaries, fallopian tubes, surrounding lymph nodes, and omentum (fatty tissue in the abdomen). After recovery, she would begin six rounds of chemotherapy with the medicines carboplatin and paclitaxel.
After her surgery, the pathology revealed her cancer was stage 3C1 and that her tumor was a certain biomarker called mismatch repair-deficient (dMMR). dMMR describes cancers that have an MMR system that is not working properly to correct errors in the genes. dMMR is an important biomarker in endometrial cancer.
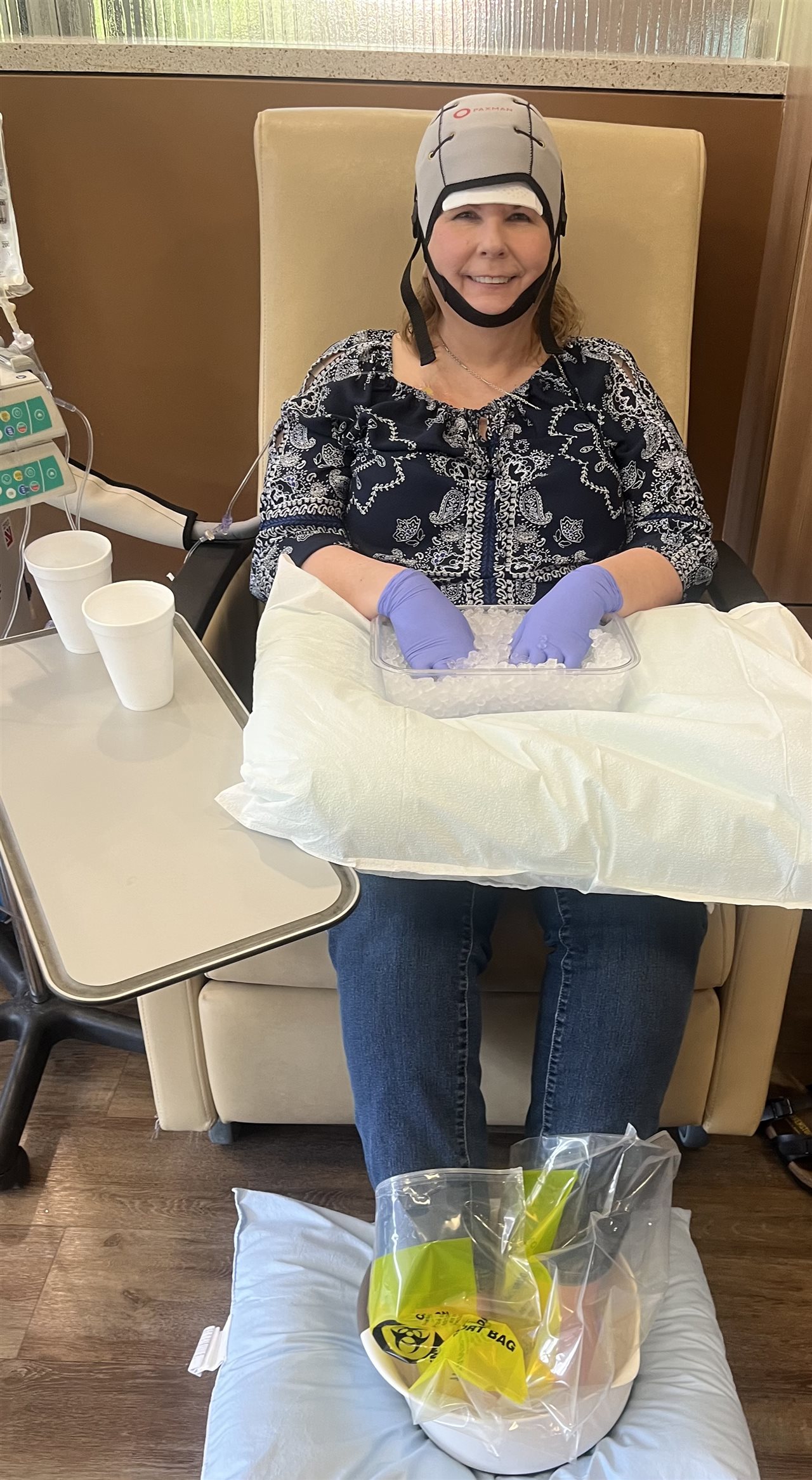
Her doctor also introduced another treatment option that they thought she could be a good candidate for: an immunotherapy called JEMPERLI (dostarlimab-gxly for injection 500 mg). JEMPERLI is an immunotherapy designed to work with the body's immune system to help find and attack cancer cells.
Approved Uses
JEMPERLI is a prescription medicine used to treat adults with a kind of uterine cancer called endometrial cancer (EC)
- JEMPERLI may be used in combination with the chemotherapy medicines, carboplatin and paclitaxel, and then after that JEMPERLI may be used alone:
- when your cancer has spread outside your uterus (advanced) or,
- your cancer has returned.
- JEMPERLI may be used alone:
- when a laboratory test shows that your tumor is mismatch repair deficient (dMMR), and
- your cancer has returned, or it has spread (advanced EC), and
- you have received chemotherapy that contains platinum and it did not work or is no longer working, and
- your cancer cannot be treated by surgery or radiation.
- It is not known if JEMPERLI is safe and effective in children.
JEMPERLI, in combination with the chemotherapy medicines carboplatin and paclitaxel, is the first and only FDA-approved immunotherapy combination proven to help patients with newly diagnosed advanced or returned endometrial cancer live longer.
A clinical trial compared JEMPERLI + CP (carboplatin and paclitaxel) vs CP alone in 494 people with endometrial cancer that had spread outside the uterus (newly diagnosed advanced) or returned, also known as the overall study population. The overall study population included people whose tumors were either dMMR/MSI-H (microsatellite instability-high) or MMRp/MSS (mismatch repair proficient)/(microsatellite stable). The clinical trial tested the effectiveness and safety of adding JEMPERLI to CP. The trial treated some patients with JEMPERLI + CP and others with a placebo (an inactive substance) + CP and then compared the results.
In the overall study population, median overall survival (OS) with JEMPERLI + CP was 45 months vs 28 months with CP alone. This difference is statistically significant, which means that the difference between the two treatment groups is greater than what might be expected by chance alone. Results varied among different biomarker groups within the overall population. Median is the middle value in a set of measurements. OS measures the average length of time patients are alive after the start of treatment. JEMPERLI + CP was not studied in comparison to other immunotherapy combinations.
Based on the results of this trial, this combination of medications was approved by the U.S. Food and Drug Administration (FDA) for healthcare providers to prescribe to appropriate patients. In patients receiving JEMPERLI + CP, 19% stopped taking JEMPERLI completely and 37% had their treatment interrupted due to side effects.
Immunotherapy like JEMPERLI may be used in certain people with advanced or recurrent endometrial cancer whose tumors have specific characteristics, such as dMMR. Based on those results, her oncologist believed JEMPERLI was right for her.
Schari'sdoctor discussed with her how JEMPERLI could affect her when reviewing treatment options. Because JEMPERLI is an immunotherapy, it can cause someone's immune system to attack normal organs and tissues in any area of the body. These problems can become severe or life-threatening and lead to death. More than one of these problems can occur at the same time, which may happen during or after treatment. It is important to call or see a doctor right away for any new or worsening signs or symptoms. Immune system problems can include lung problems, intestinal problems, liver problems, hormone gland problems, kidney problems, skin problems, and problems in other organs and tissues. JEMPERLI can also cause severe orlife-threateninginfusion reactions, rejection of a transplanted organ, and complications in people who received a bonemarrow transplant that uses donor stem cells.
The most common side effects of JEMPERLI when given with carboplatin and paclitaxel include nerve problems in your arms, hands, legs, and feet; tiredness; nausea; hair loss; joint pain; rash; constipation; diarrhea; stomach-area (abdomen) pain; shortness of breath; decreased appetite; urinary tract infections; vomiting.
These are not all the potential side effects that can occur with JEMPERLI; please review the Important Safety Information below and the fullPrescribing Information, includingMedication Guide, to learn more. Learn more about additional trial results at JEMPERLI.com and ask your doctor if JEMPERLI may be right for you or a loved one.
After completing six rounds of chemotherapy plus JEMPERLI, she transitioned to JEMPERLI alone. Every six weeks, she receives a 30-minute infusion. She's experienced some fatigue and headaches. She works closely with her doctor on her treatment schedule, including anyadditional appointments for monitoring.Maintaining this coordination is key.
Over time, her scans showed she was in remission, or what they call no evidence of disease-meaning theydon't see any cancer in her body-words she never imagined she would hear after such an advanced diagnosis. She continues to bemonitored by her doctor and is still receiving JEMPERLI.
This wasSchari's experience, and everyone's experience is different. But the results gave her something shehadn't felt in a long time: hope.
Balancing Urgency With Hope
Schari is clear about what she wants other women to take from her experience: urgency, without fear. "I often say to other women: Don't ignore those 'womanly' symptoms. I carried extra weight for years and brushed off bleeding and other signs that I now know mattered. I wish I'd been more proactive. If that's happening to you, do something now, talk to your doctor."
At the same time, her story offers hope. Advances in genetic testing, targeted therapies, and immunotherapy are expanding what's possible for people diagnosed with endometrial cancer, even at later stages.
"I used to believe that if cancer ever came for me, I wouldn't be able to handle it. However, here I am, battle-weary, but still standing. It hasn't been easy. My life is forever changed. Now I live with a new reality: the beast called cancer still lurks at the edge of the woods, always watching, always waiting. But I no longer face it empty-handed. And for that, I am thankful."
Her journey is a reminder that awareness can lead to earlier evaluation, that science is reshaping treatment, and that even when diagnosis comes late, progress in care is giving patients and families something powerful to hold onto: possibility.
To learn more about endometrial cancer and potential treatment options, including JEMPERLI, talk to your doctor and visitwww.JEMPERLI.com.
IMPORTANT SAFETY INFORMATION
JEMPERLI is a medicine that may treat certain cancers by working with your immune system. JEMPERLI can cause your immune system to attack normal organs and tissues in any area of your body and can affect the way they work. These problems can sometimes become severe or life-threatening and can lead to death. You can have more than one of these problems at the same time. These problems may happen anytime during treatment or even after your treatment has ended.
Call or see your healthcare provider (HCP) right away if you develop any new or worsening signs or symptoms, including:
Lung problems. Signs and symptoms may include cough, shortness of breath, or chest pain.
Intestinal problems. Signs and symptoms may include diarrhea or more bowel movements than usual; stools that are black, tarry, sticky, or have blood or mucus; or severe stomach-area (abdomen) pain or tenderness.
Liver problems. Signs and symptoms may include yellowing of your skin or the whites of your eyes, severe nausea or vomiting, pain on the right side of your stomach area (abdomen), dark urine (tea colored), or bleeding or bruising more easily than usual.
Hormone gland problems. Signs and symptoms may include headaches that will not go away or unusual headaches, eye sensitivity to light, eye problems, rapid heartbeat, increased sweating, extreme tiredness, weight gain or weight loss, feeling more hungry or thirsty than usual, urinating more often than usual, hair loss, feeling cold, constipation, your voice gets deeper, dizziness or fainting, changes in mood or behavior such as decreased sex drive, irritability, or forgetfulness.
Kidney problems. Signs and symptoms may include change in the amount or color of your urine, blood in your urine, swelling in your ankles, or loss of appetite.
Skin problems. Signs and symptoms may include rash; itching; skin blistering or peeling; swollen lymph nodes; painful sores or ulcers in your mouth or in your nose, throat, or genital area; fever or flu-like symptoms.
Problems can also happen in other organs and tissues. These are not all of the signs and symptoms of immune system problems that can happen with JEMPERLI. Call or see your HCP right away for any new or worse signs or symptoms. Signs and symptoms may include chest pain, irregular heartbeat, shortness of breath, swelling of ankles; confusion, sleepiness, memory problems, changes in mood or behavior, stiff neck, balance problems, tingling or numbness of the arms or legs; double vision, blurry vision, sensitivity to light, eye pain, changes in eyesight; persistent or severe muscle pain or weakness, muscle cramps; low red blood cells, bruising.
Infusion reactions that can sometimes be severe or life-threatening. Signs and symptoms of infusion reactions may include chills or shaking, itching or rash, flushing, shortness of breath or wheezing, dizziness, feel like passing out, fever, back or neck pain.
Rejection of a transplanted organ. Your HCP should tell you what signs and symptoms you should report and monitor you, depending on the type of organ transplant that you have had.
Complications, including graft-versus-host-disease (GVHD), in people who have received a bone marrow (stem cell) transplant that uses donor stem cells (allogeneic). These complications can be serious and can lead to death. These complications may happen if you underwent transplantation either before or after being treated with JEMPERLI. Your HCP will monitor you for these complications.
Getting medical treatment right away may help keep these problems from becoming more serious. Your HCP will check you for these problems during treatment with JEMPERLI and may treat you with corticosteroid or hormone replacement medicines. If you have severe side effects, your HCP may also need to delay or completely stop treatment with JEMPERLI.
Before receiving JEMPERLI, tell your HCP about all of your medical conditions, including immune system problems such as Crohn's disease, ulcerative colitis, or lupus; received an organ transplant; have received or plan to receive a stem cell transplant that uses donor stem cells (allogeneic); have received radiation treatment to your chest area; have a condition that affects your nervous system, such as myasthenia gravis or Guillain-Barré syndrome.
If you are pregnant or plan to become pregnant, tell your HCP. JEMPERLI can harm your unborn baby. If you are able to become pregnant, your HCP will give you a pregnancy test before you start treatment. Use an effective birth control method during treatment and for 4 months after your last dose of JEMPERLI. Tell your HCP right away if you become pregnant or think you may be pregnant during treatment with JEMPERLI.
If you are breastfeeding or plan to breastfeed, tell your HCP. It is not known if JEMPERLI passes into your breast milk. Do not breastfeed during treatment with JEMPERLI and for 4 months after your last dose.
Tell your HCP about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
The most common side effects of JEMPERLI when given with carboplatin and paclitaxel include nerve problems in your arms, hands, legs, and feet; tiredness; nausea; hair loss; joint pain; rash; constipation; diarrhea; stomach-area (abdomen) pain; shortness of breath; decreased appetite; urinary tract infections; vomiting.
The most common side effects of JEMPERLI when used alone include tiredness and weakness, low red blood cell count (anemia), diarrhea, nausea, constipation, vomiting.
These are not all of the possible side effects of JEMPERLI. Call your doctor for medical advice about side effects.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch or call by 1-800-FDA-1088. You may also report negative side effects to GSK at https://gsk.public.reportum.com or 1-888-825-5249.
Please see fullPrescribing Information, includingMedication Guide for patients.
Trademarks are owned by or licensed to the GSK group of companies.
©2026 GSK or licensor.
PMUS-DSTCOCO260002 April 2026
Produced in USA.
Source: BrandPoint





















