Theralase(R) Demonstrates That X-Ray-Activated Rutherrin(R) Is Significantly More Effective than Radiation Alone in the Destruction of Muscle Invasive Bladder Cancer
Toronto, Ontario--(Newsfile Corp. - March 9, 2026) - Theralase® Technologies Inc. (TSXV: TLT) (OTCQB: TLTFF) ("Theralase®" or the "Company"), a clinical stage pharmaceutical company dedicated to the research and development of energy-activated small molecules for the safe and effective destruction of cancer, bacteria and viruses, is pleased to present its latest preclinical data demonstrating that X-Ray-activated Rutherrin® is effective in the destruction of Muscle Invasive Bladder Cancer ("MIBC").
In the MIBC preclinical models conducted by the Theralase® research team, Rutherrin® demonstrated an ability to significantly enhance the efficacy of radiation therapy in the destruction of MIBC, supporting its potential as an effective drug for this devastating condition.
Approximately 25% of the 83,000+ new bladder cancer cases diagnosed annually in the United States and 13,300+ cases in Canada are MIBC. MIBC carries a significant risk of death that has not changed in decades.1,2
The standard of care for MIBC is currently cisplatin-based neoadjuvant chemotherapy (pre-operative) followed by Radical Cystectomy ("RC") (bladder removal).1 In patients who undergo RC, systemic recurrence rates vary by stage, ranging from 20% to 70%.3
As a bladder-preservation option, Tri Modal Therapy ("TMT"), comprised of maximal trans-urethral resection of the bladder tumour, followed by radiation and chemotherapy, has become increasingly popular in recent years; however, patients must be appropriately selected.4,5
Based on the latest preclinical data, Theralase® is planning to conduct a Phase 0/I/II adaptive clinical study with radiation-activated Rutherrin®, with or without chemotherapy and/or immunotherapy, subject to regulatory approval, for patients diagnosed with MIBC to provide an alternative treatment option versus RC or TMT.
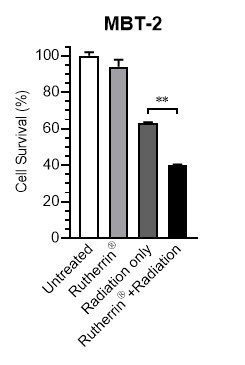
In vitro experiments using mouse MIBC cells ("MBT-2"), Rutherrin® combined with 2.5 Gray X-Ray radiation resulted in significantly greater cell death compared to radiation alone (Figure 1).
These results indicate that Rutherrin® enhances radiation-induced cytotoxicity, supporting its potential as a radiosensitizer in the destruction of MIBC.
Figure 1: In Vitro MBT-2 Cell Kill After Rutherrin® and 2.5 Gray X-Ray Treatment
To view an enhanced version of this graphic, please visit:
https://images.newsfilecorp.com/files/2786/286595_17f6f13c641d1bf2_001full.jpg
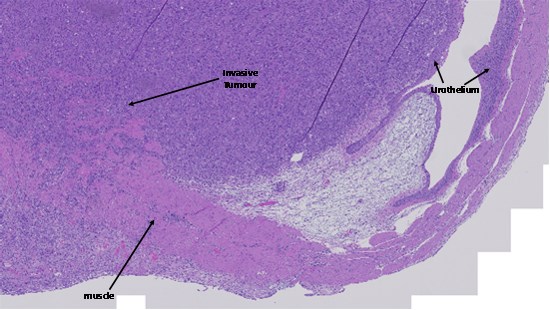
MBT-2 cells were used to establish an orthotopic MIBC model in mice, by surgically injecting the cells directly into the muscle layer of the mouse's bladder, leading to tumour growth and invasion of the muscle layer. In this model, the tumour grew into the bladder lumen, pushing against the inner urothelial layer, resulting in a reduction of the volume available for urine storage (Figure 2). If left untreated, the tumour would continue to grow until the bladder was completely blocked, rendering it inoperable and reaching the humane endpoint.
Figure 2: Histological Analysis Confirmed Successful Establishment of a MIBC Model
To view an enhanced version of this graphic, please visit:
https://images.newsfilecorp.com/files/2786/286595_17f6f13c641d1bf2_002full.jpg
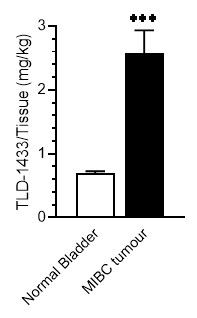
After systemic Rutherrin® was administered intravenously to mice with MBT-2 tumours, tissue analysis showed that tumour sites had approximately four times higher Rutherrin® levels than normal bladder, demonstrating tumour-selective uptake and a favourable therapeutic index (drug in cancer cells versus healthy cells). See Figure 3.
Figure 3: Selective Tumour Uptake of Rutherrin®
To view an enhanced version of this graphic, please visit:
https://images.newsfilecorp.com/files/2786/286595_17f6f13c641d1bf2_003full.jpg
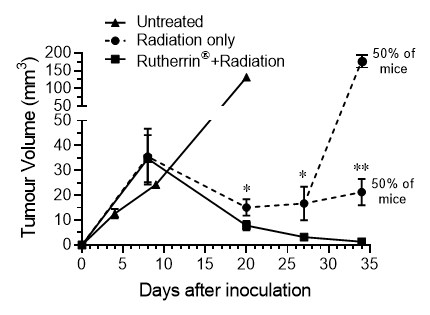
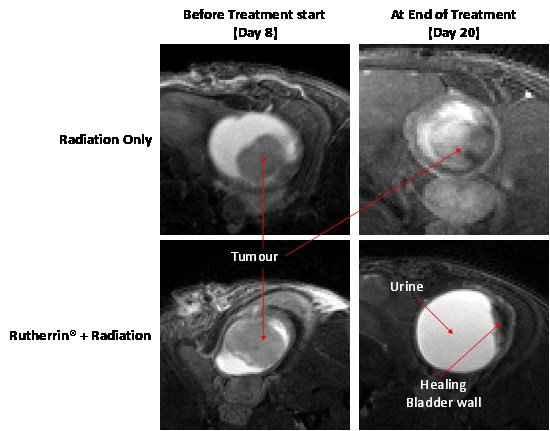
In further research, Theralase® is conducting an ongoing in-vivo MIBC study assessing the use of repeatable, intravenous Rutherrin® administration, in combination with fractionated radiation therapy (total cumulative dose of 25 Gray). At day 35 in this study, all mice in the Rutherrin® + radiation group survived and showed complete tumour clearance; however, in the radiation alone group mice exhibited persistent and larger tumour volumes, with 50% of the mice reaching their humane endpoint post treatment, with the other 50% displaying steady growth in their tumours. (Figure 4 and 5). These results indicate that Rutherrin® significantly enhanced radiation efficacy and may improve tumour control even when lower doses of radiation are employed.
Figure 4: Tumour Volume Analysis in MBT-2 Mouse Model Treated with 25 Gray Cumulative Radiation
To view an enhanced version of this graphic, please visit:
https://images.newsfilecorp.com/files/2786/286595_17f6f13c641d1bf2_004full.jpg
Figure 5: Representative MRI Sections of the Mouse Bladder Before and After Treatments
To view an enhanced version of this graphic, please visit:
https://images.newsfilecorp.com/files/2786/286595_17f6f13c641d1bf2_005full.jpg
Mark Roufaiel, PhD, Research Scientist at Theralase®, commented, "These preclinical findings demonstrate that Rutherrin® selectively accumulates in MIBC tumours versus health bladder tissue and significantly enhanced the therapeutic effect of radiation. Most importantly, we observed complete tumour clearance in X-Ray-activated Rutherrin® treated animals even under low radiation dosing conditions. This supports the potential of Rutherrin® to improve radiation response and expand treatment options for patients with aggressive MIBC."
Arkady Mandel, MD, PhD, DSc, Chief Scientific Officer, Theralase®, stated, "Treatment options for MIBC patients are limited to cisplatin-based chemotherapy before RC or TMT. Preclinical data collected supports the clinical opportunity of employing X-Ray-Activated Rutherrin® in the treatment of difficult-to-treat cancers; such as, glioblastoma multiforme, non-small cell lung cancer, as previously reported, and now based on the recent data, MIBC. In preclinical studies, mice treated with radiation alone did not survive or continued to show detectable cancer with rapid tumour progression; whereas, mice treated with a combination of Rutherrin® and radiation therapy showed no detectable cancer. These results will hopefully lay the groundwork for new therapeutic approaches for patients faced with these challenging diseases."
Roger DuMoulin-White, BSc, P.Eng, Pro.Dir, President, Chief Executive Officer and Chairman of the Board of Theralase®, added, "The combination of selective tumour uptake by Rutherrin®, demonstrated X-Ray enhancement in vitro, accelerated tumour regression and complete tumour destruction in vivo at low radiation doses collectively supports the executive decision by Theralase® to pursue clinical development of Rutherrin®, as a radiation sensitizer in patients diagnosed with GBM, NSCLC and MIBC, subject to regulatory approval."
References:
1 Patel M et al. Bladder sparing management for muscle-invasive bladder cancer after a complete clinical response: ready for prime time? -a narrative review. Transl Cancer Res. 2024 Nov 30;13(11):6413-6429.
2 Kulkarni et al. 2025 Canadian Urological Association Guideline: Muscle-invasive bladder cancer. Can Urol Assoc J. 2025;19(I):E1-16.
3 Holzbeierlein J et al. Treatment of Non-Metastatic Muscle-Invasive Bladder Cancer: AUA/ASCO/SUO Guideline (2017; Amended 2020, 2024). Journal of Urology [Internet]. 2024 Jul 1. 212(1):3-10.
4 Su X et al. Oncological effectiveness of bladder-preserving trimodal therapy versus radical cystectomy for the treatment of muscle-invasive bladder cancer: a system review and meta-analysis. World J Surg Oncol. 2023 Aug 29;21(1):271.
5 Embracing Trimodal Therapy as a Viable Alternative to Radical Cystectomy. Uro Today. May 2024
About Theralase® Technologies Inc.:
Theralase® is a clinical stage pharmaceutical company dedicated to the research and development of energy-activated small molecules for the safe and effective destruction of cancer, bacteria and viruses.
Additional information is available at www.theralase.com and www.sedarplus.ca.
Neither TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
Forward-Looking Statements
This news release contains Forward-Looking Statements ("FLS") within the meaning of applicable Canadian securities laws. Such statements include; but, are not limited to statements regarding the Company's proposed development plans with respect to small molecules and their drug formulations. FLS may be identified by the use of the words "may", "should", "will", "anticipates", "believes", "plans", "expects", "estimate", "potential for" and similar expressions; including, statements related to the current expectations of the Company's management regarding future research, development and commercialization of the Company's small molecules; their drug formulations; preclinical research; clinical studies and regulatory approvals.
These statements involve significant risks, uncertainties and assumptions; including, the ability of the Company to fund and secure regulatory approvals to successfully complete various clinical studies in a timely fashion and implement its development plans. Other risks include: the ability of the Company to successfully commercialize its small molecule and drug formulations; access to sufficient capital to fund the Company's operations is available on terms that are commercially favorable to the Company or at all; the Company's small molecule and formulations may not be effective against the diseases tested in its clinical studies; the Company fails to comply with the terms of license agreements with third parties and as a result loses the right to use key intellectual property in its business; the Company's ability to protect its intellectual property; the timing and success of submission, acceptance and approval of regulatory filings. Many of these factors that will determine actual results are beyond the Company's ability to control or predict.
Readers should not unduly rely on these FLS, which are not a guarantee of future performance. There can be no assurance that FLS will prove to be accurate as such FLS involve known and unknown risks, uncertainties and other factors, which may cause actual results or future events to differ materially from the FLS.
Although the FLS contained in the press release are based upon what management currently believes to be reasonable assumptions, the Company cannot assure prospective investors that actual results, performance or achievements will be consistent with these FLS.
All FLS are made as of the date hereof and are subject to change. Except as required by law, the Company assumes no obligation to update such FLS.
For investor information on the Company, please feel to reach out Investor Inquiries - Theralase Technologies.

To view the source version of this press release, please visit https://www.newsfilecorp.com/release/286595
Source: Theralase Technologies Inc.
© 2026 Newsfile Corp. All rights reserved.