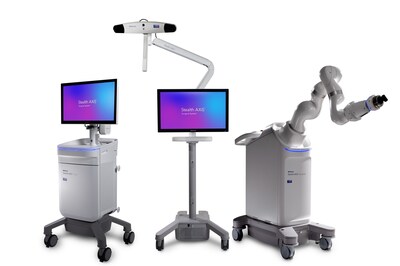
Medtronic announces CE Mark for Stealth AXiS™ surgical system
Medtronic announces CE Mark for Stealth AXiS™ surgical system |
| [28-April-2026] |
Following recent FDA clearances, Medtronic is accelerating access to integrated planning, navigation, and robotics platform across Europe GALWAY, Ireland, April 28, 2026 /PRNewswire/ -- Medtronic, a global leader in healthcare technology, has received CE mark for the Stealth AXiS™ surgical system, a next-generation platform that brings planning, navigation, and robotics together into one single, intelligent system. The Stealth AXiS™ system is indicated for spine and cranial procedures, highlighting the platform's scalability across multiple surgical specialties for Medtronic. "Receiving CE mark for the Stealth AXiS™ system marks an important milestone in expanding access to advanced surgical technologies across Europe," said Oli Prosperi, Vice President, International Commercial, Cranial & Spinal Technologies, Medtronic. "Achieving CE mark in the current regulatory environment, under EU Medical Device Regulation (MDR), underscores our commitment to bringing meaningful innovation into a highly diverse healthcare landscape." The Stealth AXiS™ system represents major advancement in surgical technology by integrating planning, navigation, and robotic execution into a single, modular platform. Designed to support a wide range of clinical workflows and surgical environments, the system enables hospitals to adopt capabilities based on their needs, while maintaining flexibility for future expansion. A key innovation of the system is LiveAlign™ segmental tracking, an industry-first capability that allows surgeons to visualize anatomical motion and patient alignment in real time during spine procedures. As a cornerstone of Medtronic's AiBLE™ ecosystem, the Stealth AXiS™ system connects pre-, intra-, and post-operative workflows, enabling a more integrated approach to surgical care. A key strength of the platform lies in its AI-driven architecture, which enables advanced planning and visualization across all stages of surgery - before, during, and after the procedure. In cranial applications, it features AI-powered automatic tractography, allowing surgeons to create patient-specific brain maps and clearly visualize critical neural pathways linked to essential brain functions. "The introduction of the Stealth AXiS™ system to Europe reflects our focus on delivering connected platforms that improve precision, efficiency, and consistency across diverse healthcare systems," said Michael Carter, Senior Vice President and President of Cranial & Spinal Technologies at Medtronic. "By bringing planning, navigation, and robotic guidance into a unified workflow, we are helping surgeons translate insight into action in the operating room, supporting more predictable and personalized care while building a strong foundation for continued innovation." The CE mark follows U.S. FDA clearances earlier this year, highlighting Medtronic's ability to rapidly bring advanced surgical technologies to global markets. About the Cranial & Spinal Technologies Business at Medtronic About Medtronic Any forward-looking statements are subject to risks and uncertainties such as those described in Medtronic's periodic reports on file with the Securities and Exchange Commission. Actual results may differ materially from anticipated results. Contacts: Ingrid Goldberg
SOURCE Medtronic plc | ||
Company Codes: NYSE:MDT |